Transport Proteins
The short intro I did in class is found here.
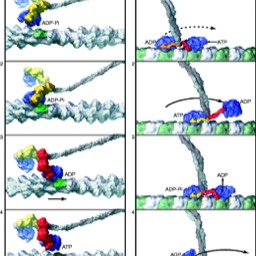
One of the main things a membrane has to be able to do is control what flows through it. Sometime, that's just opening a channel and letting the dissolved thing flow "down gradient." But, sometime the cell has to do active work to pump ions to one side or the other of a membrane.
Once that gradient is established, it can be used to do work…and in fact used in myriad ways you can hardly imagine. Since it is so important, we will spend some time on it.
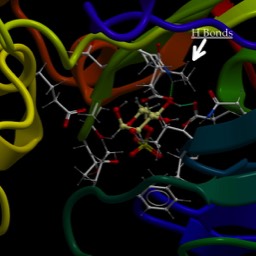
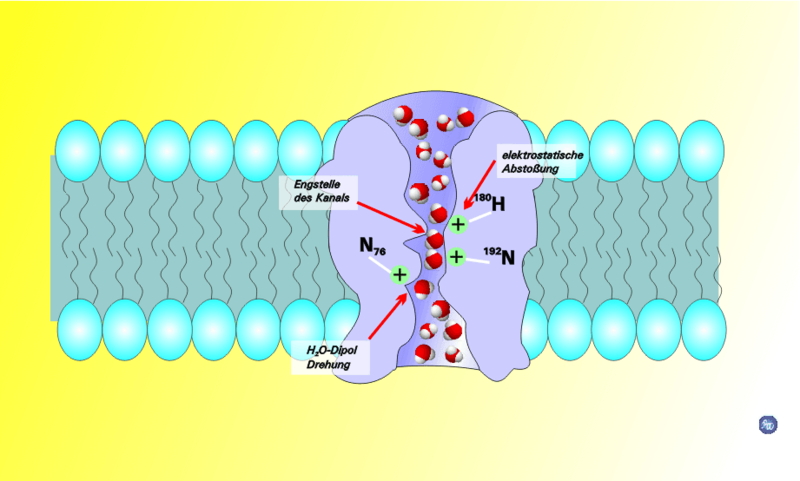
By the way, the figure is a cartoon of a protein called "aquaporin." Guess what it transports. It's a fascinating protein. Think about how you can make a protein that allows water…and only water…to pass through.
One of the main things a membrane has to be able to do is control what flows through it. Sometime, that's just opening a channel and letting the dissolved thing flow "down gradient." But, sometime the cell has to do active work to pump ions to one side or the other of a membrane.
Once that gradient is established, it can be used to do work…and in fact used in myriad ways you can hardly imagine. Since it is so important, we will spend some time on it.
By the way, the figure is a cartoon of a protein called "aquaporin." Guess what it transports. It's a fascinating protein. Think about how you can make a protein that allows water…and only water…to pass through.