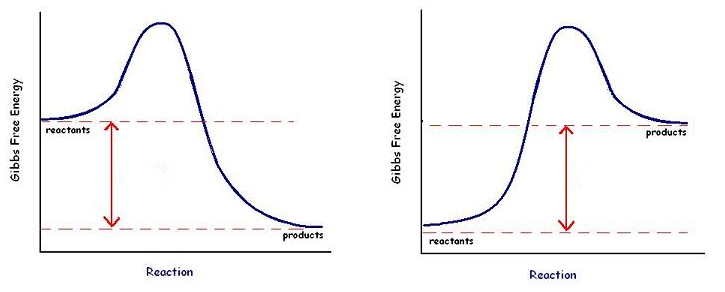
Kinetics Pogil Answers
Here is a link to a practice worksheet on kinetics.Pogil Answers.
Here is a link to the answer keys for the Pogil activities. First one is ready now.
First Pogil Answers. Note that the answers should be good…but, if you have different answers, don't be concerned. I make mistakes.
Here is the second one.
Also, here are some reaction mechanism problems:
Below is an overall reaction and a proposed reaction mechanism:
Overall:
2NO2Cl(g) → 2NO2(g) + Cl2(g)
- NO2Cl → NO2 + Cl (slow)
- NO2Cl + Cl → NO2 + Cl2 (fast)
Assuming the reaction mechanism above is correct
- What is the rate determining step?
- What should be the rate law for the overall reaction?
- What reaction intermediate is present?
Answers below:
The rate determining (limiting) step is the first one. That is, the slow one.
The rate law should be Rate=k[NO2Cl]
The reaction intermediate is Cl a chlorine atom.